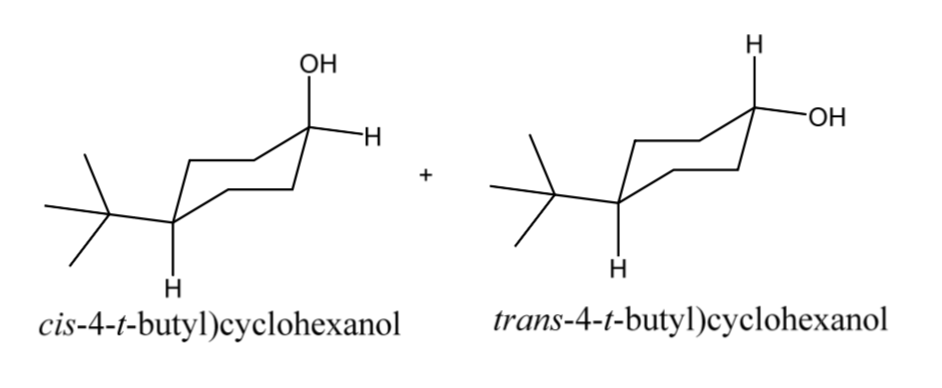
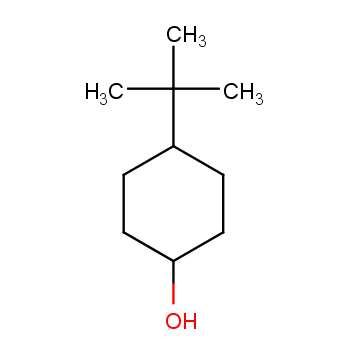
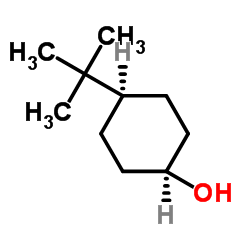
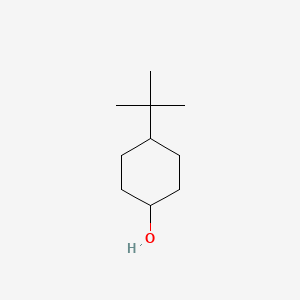
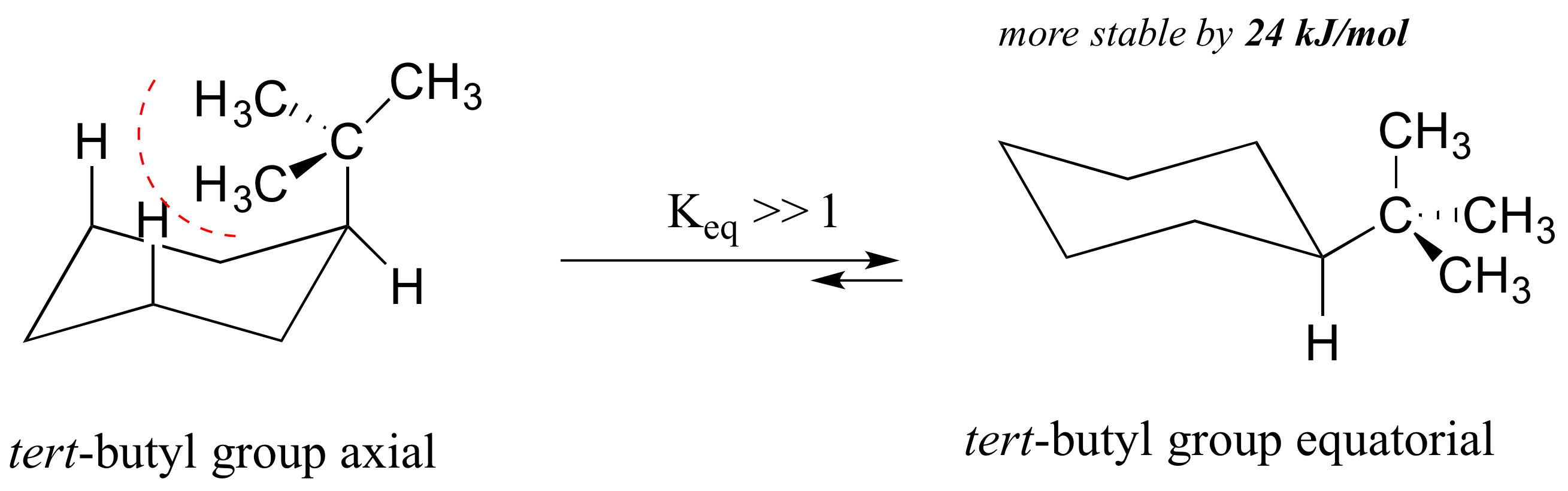
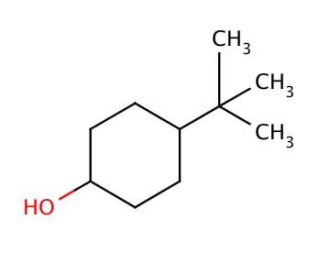
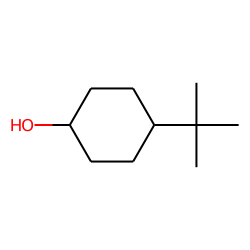
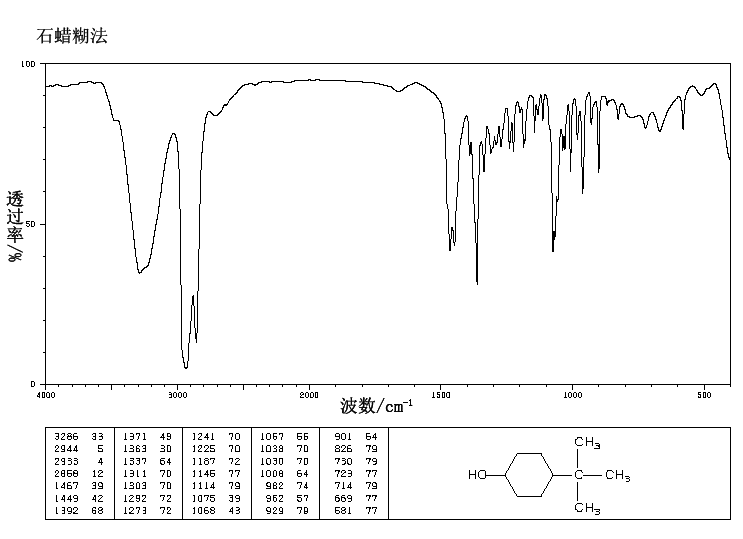
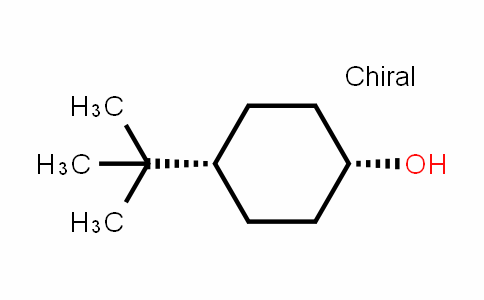
4-tert-Butylcyclohexanol (Cis/trans ratio: approximately 70% trans, 30% cis) | 98-52-2 | Manchester Organics

SOLVED: Oxidation of 4-tert-butylcyclohexanol by sodium hypochlorite produces 4-tert-butylcyclohexanone (shown below): If you were asked to monitor the progress of the reaction by think layer chromatography, the Rf value of the product (